Reducing anticholinergic burden to improve safety
September 15, 2022

 Contributed by:
Contributed by:
Cheryl A Sadowski, B.Sc.(Pharm), Pharm.D., FCSHP - View bio
Case
Mrs. R is an 81-year-old widow who lives with her daughter and her family. She was referred to the geriatric outpatient clinic for assessment of cognition and mobility. She recently fell at home and has had pain in her back and legs since that time. The family also noticed recently that Mrs. R has been paying less attention to conversation during meals, becomes confused when asked questions, has been losing her glasses because she takes them off frequently and has been coughing frequently (which is new). She also describes new abdominal pain. Her medical history and treatments are below:
|
Condition |
Medication |
Comment |
|
Depression |
Venlafaxine 75mg daily |
Started five years ago. |
|
Insomnia |
Sleep aid (diphenhydramine 50mg, acetaminophen 325 mg) QHS |
Started one month ago. Recently switched from melatonin after she read an article related to melatonin safety. |
|
Hypertension |
Amlodipine 10 mg daily |
Started eight years ago. |
|
Reflux, occasionally |
Ranitidine OTC 150 mg BID PRN |
Patient uncertain when started. |
|
Constipation |
Senna 8.6 mg, 2 tabs QHS PRN |
Started two weeks ago. |
|
Hypothyroidism |
Levothyroxine 75 mcg daily |
Started on levothyroxine over 20 years ago. |
|
Overactive bladder |
Solifenacin 5 mg daily |
Started four years ago. |
|
Osteoarthritis |
Ibuprofen 200 mg TID PRN |
Patient uncertain when started. |
|
Back pain |
Cyclobenzaprine 10 mg QHS |
Started one month ago. |
Question
What can be done to reduce anticholinergic burden to improve safety in older adults?
Background
Greek mythology helps to explain the potential severity of anticholinergic medications. Atropine, one of the most potent, CNS-penetrating anticholinergic medications available, is named after the Greek Fate, Atropos, who was responsible for cutting the thread of life, determining the length of life for each human.1
While anticholinergic medications are a distinct class within the WHO Anatomic Therapeutic Chemical Classification, including medications such as trihexyphenidyl, most medications with anticholinergic effects are distributed in other classes, such as antihistamines, anti-seizure medication or antidepressants. There are also classes of medications that are anticholinergic, but selectively. The anti-muscarinic treatments used in overactive bladder are a good example of targeting a particular muscarinic receptor. In this case the muscarinic receptors in the bladder wall are selectively blocked, with minimal effect on other muscarinic receptors throughout the body, leading to fewer complaints of xerostomia, constipation or confusion.2
The side effects can impact almost every organ system. Secretions dry up, swallowing can be more difficult with xerostomia and can even lead to dysphagia, eyes become dry, gut motility is reduced, urine is retained and pupils may not respond to light appropriately.3 Geriatric syndromes that can be affected include falls, frailty, functional decline, delirium and cognitive decline.4-9 A higher burden of anticholinergic medications is associated with lower quality of life.10 Even mortality and cardiovascular events are increased with exposure to anticholinergics.11 Older adults are particularly susceptible to these side effects due to changes in pharmacodynamics and pharmacokinetics that occur with aging. These changes lead to greater sensitivity to anticholinergic effects, leading to toxicity at even low doses.12
Preventing anticholinergic toxicity can be challenging because the effects can accumulate from a number of medications, and many of the medications are over-the-counter (OTC), resulting in gaps in the medication records that are typically consulted. Diphenhydramine, dimenhydrinate, scopolamine, sleep aids with antihistamines or even analgesic combinations with muscle relaxants are all available OTC. Adding up the effects of anticholinergics can be done with tools. There is not one universally accepted scale but validated resources such as the Anticholinergic Cognitive Burden Scale or Drug Burden Index can provide scoring or a weight related to the anticholinergic potency of each medication, and the AGS Beers Criteria or STOPP criteria can be consulted to identify anticholinergic medications that should be discontinued.13-16
In Canada anticholinergic medication use is common, although precise prevalence is uncertain due to OTC medications. Almost half of older adults receive a claim for an AGS Beers Criteria medication each year, medications that are deemed to have more harm than benefit in older adults, and for which other safer treatments are available. These criteria are used by the Canadian Institute of Health Information (CIHI) and other international agencies, as criteria for potentially inappropriate medications for the population that is age 65 years and older. Many of the medications in the AGS Beers Criteria are either sedating and/or anticholinergic.17
Recommendation
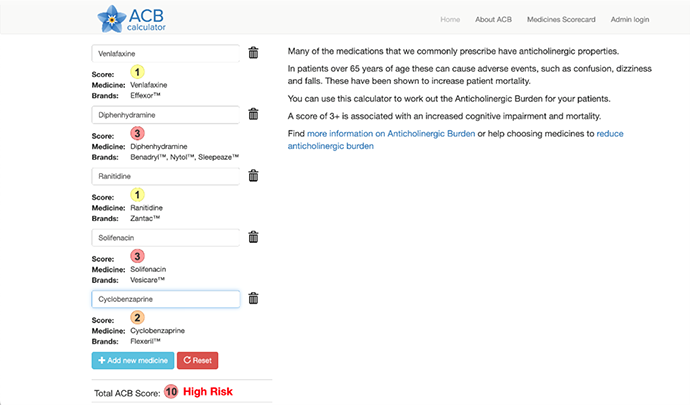
Returning to the patient case, we can see that this patient has had an accumulation of a few anticholinergic medications.13 Diphenhydramine, often used as an OTC sleep aid, is a potent anticholinergic. The ACB tools ranks this as the highest score, 3/3 for anticholinergic potency. Solifenacin is also ranked as 3/3 for anticholinergic effect, but it is a selective antimuscarinic which is less likely to be causing CNS effects, but peripherally it may lead to urinary retention when combined with other anticholinergics. Cyclobenzaprine is a moderate anticholinergic, scored as 2/3 for anticholinergic effect, while both ranitidine and venlafaxine have mild anticholinergic effects, both 1/3 for anticholinergic effect. Overall this patient has been exposed to quite a significant anticholinergic load, likely leading to her constipation, dry mouth and resulting cough and confusion.
An example of how to use on line calculators can be seen here: www.acbcalc.com
Clinicians can improve medication safety by becoming aware of which medications have anticholinergic effects,18 asking specifically about high-risk medications (e.g., “Do you use an over-the-counter sleep aid? Do you use a product like BenadrylⓇ?) and discontinuing medications or switching to a similar medication that has less anticholinergic effect. A team approach using comprehensive geriatric assessment and inquiry about changes in function, cognition or side effects associated with new medications can also identify high risk cases.14
References
- Holzman Robert S. The Legacy of Atropos, the Fate Who Cut the Thread of Life Anesthesiology. 1998;89(1):241-9.
- Wagg AS. Antimuscarinic treatment in overactive bladder: special considerations in elderly patients. Drugs & aging. 2012;29(7):539-48.
- Pierce H, Thomas D, Asfaw T, Chughtai B. Anticholinergic Burden in the Elderly Population: An Emerging Concern. European urology. 2019;76(1):7-8.
- Landi F, Dell'Aquila G, Collamati A, Martone AM, Zuliani G, Gasperini B, et al. Anticholinergic drug use and negative outcomes among the frail elderly population living in a nursing home. J Am Med Dir Assoc. 2014;15(11):825-9.
- Green AR, Reifler LM, Bayliss EA, Weffald LA, Boyd CM. Drugs Contributing to Anticholinergic Burden and Risk of Fall or Fall-Related Injury among Older Adults with Mild Cognitive Impairment, Dementia and Multiple Chronic Conditions: A Retrospective Cohort Study. Drugs & Aging. 2019;36(3):289-97.
- Richardson K, Fox C, Maidment I, Steel N, Loke YK, Arthur A, et al. Anticholinergic drugs and risk of dementia: case-control study. Bmj. 2018;361:k1315.
- Coupland CAC, Hill T, Dening T, Morriss R, Moore M, Hippisley-Cox J. Anticholinergic Drug Exposure and the Risk of Dementia: A Nested Case-Control Study. JAMA Intern Med. 2019;179(8):1084-93.
- Gerretsen P, Pollock BG. Cognitive risks of anticholinergics in the elderly. Aging Health. 2013;9(2):159-66.
- Naharci MI, Tasci I. Frailty status and increased risk for falls: The role of anticholinergic burden. Archives of Gerontology & Geriatrics. 2020;90:104136.
- Stewart C, Yrjana K, Kishor M, Soiza RL, Taylor-Rowan M, Quinn TJ, et al. Anticholinergic Burden Measures Predict Older People's Physical Function and Quality of Life: A Systematic Review. J Am Med Dir Assoc. 2021;22(1):56-64.
- Graves-Morris K, Stewart C, Soiza RL, Taylor-Rowan M, Quinn TJ, Loke YK, et al. The Prognostic Value of Anticholinergic Burden Measures in Relation to Mortality in Older Individuals: A Systematic Review and Meta-Analysis. Frontiers in Pharmacology. 2020;11:570.
- Zazzara, M.B., Palmer, K., Vetrano, D.L. et al. Adverse drug reactions in older adults: a narrative review of the literature. Eur Geriatr Med 12, 463–473 (2021). https://doi.org/10.1007/s41999-021-00481-9
- Gerretsen P, Pollock BG. Drugs with anticholinergic properties: a current perspective on use and safety. Expert Opinion on Drug Safety. 2011;10(5):751-65.
- Sanchini C, Negri S, Cesari M. The complexity of the anticholinergic burden in frail older persons. Advances in Geriatric Medicine and Research. 2021;3(1):e210001.
- By the 2019 American Geriatrics Society Beers Criteria® Update Expert Panel. American Geriatrics Society 2019 Updated AGS Beers Criteria® for Potentially Inappropriate Medication Use in Older Adults. J Am Geriatr Soc. 2019 Apr;67(4):674-694. doi: 10.1111/jgs.15767. Epub 2019 Jan 29. PMID: 30693946.
- O'Mahony D, O'Sullivan D, Byrne S, O'Connor MN, Ryan C, Gallagher P. STOPP/START criteria for potentially inappropriate prescribing in older people: version 2. Age Ageing. 2015 Mar;44(2):213-8. doi: 10.1093/ageing/afu145. Epub 2014 Oct 16. Erratum in: Age Ageing. 2018 May 1;47(3):489. PMID: 25324330; PMCID: PMC4339726.
- Canadian Institute for Health Information. Drug Use Among Seniors in Canada, 2016. Ottawa, ON: CIHI; 2018.
- Araklitis G, Thiagamoorthy G, Hunter J, Rantell A, Robinson D, Cardozo L. Anticholinergic prescription: are healthcare professionals the real burden? Int Urogynecol J. 2017;28(8):1249-56.
Additional Patient Resource
Anticholinergic Toxicity. Available from: www.youtube.com/watch?v=Y5atl1pWtUs
